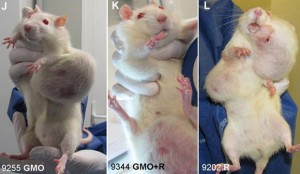
In 2012, a study by the French research group CRIIGEN made international headlines with images of laboratory rats bearing massive tumors after being fed genetically engineered corn treated with Roundup herbicide. The study reignited debates about the safety of genetically modified organisms in the food supply and drew attention to the intersection of agricultural biotechnology, government regulation, and national security policy.
While the CRIIGEN study itself would later face significant scientific criticism regarding its methodology and sample sizes, the questions it raised about regulatory oversight and corporate influence in food safety remained relevant.
Regulatory Capture and the Revolving Door
Critics of the American regulatory framework for GMOs have long pointed to the close relationship between Monsanto and the agencies responsible for food safety oversight. Michael Taylor, a former Monsanto vice president, held the position at the FDA responsible for food safety policy. The USDA had funded research into biotechnology applications including corn varieties with contraceptive properties. The EPA faced criticism for pursuing regulatory actions against small agricultural operations while allegedly overlooking more significant environmental contamination.
These connections raised concerns about whether regulatory agencies were functioning as independent watchdogs or as facilitators for the biotechnology industry. The pattern of personnel movement between Monsanto and government agencies created at minimum the appearance of a conflict of interest.
Agricultural Biosecurity and Organic Farming
A more contentious development involved the classification of certain organic farming practices under biosecurity frameworks. The Department of Homeland Security funded agricultural biosecurity networks, including at Stanford University, with the stated goal of coordinating responses to agricultural pests and pathogens that could be used in bioterrorism.
The USDA adopted positions characterizing certain aspects of organic agriculture, including free-range livestock and open-pollinated seeds, as potential biosecurity risks. Agency directives outlined approaches to containing organic farming operations in ways that critics argued would effectively convert them into factory-style operations, eliminating the very characteristics that distinguished organic agriculture.
This framing struck many observers as paradoxical. Traditional farming practices that had sustained human agriculture for millennia were being reclassified as threats, while industrial monoculture operations dependent on proprietary seeds and chemical inputs were treated as the standard of safety.
The International Dimension
The tension between biotechnology interests and food safety extended beyond American borders. In India, proposed legislation would have established a biotech regulatory authority with provisions that critics described as criminalizing public questioning of GMO safety. The bill was characterized by Indian legal scholars and scientists as unconstitutional and unscientific.
Reports from the field documented a troubling pattern: genetically modified crops promoted as high-yield solutions had in many cases failed to deliver promised productivity improvements while creating economic dependencies on proprietary seed systems. In India, the financial burden of purchasing patented seeds contributed to a documented epidemic of farmer suicides.
The Vaccine and Pharmaceutical Connection
The agricultural biotechnology debate intersected with broader concerns about pharmaceutical regulation. Several companies involved in GMO crop development also operated in the vaccine and pharmaceutical sectors. Critics pointed to this overlap as evidence of a consolidated corporate influence over multiple aspects of public health policy.
Freedom of Information requests in the United Kingdom revealed that government agencies had possessed data for decades suggesting that certain vaccines were less effective than publicly claimed and were associated with adverse health effects. In the United States, government agencies faced legal challenges for withholding FOIA documents related to vaccine safety research.
These revelations contributed to growing public skepticism about the independence of regulatory agencies and their willingness to prioritize public health over industry relationships.
The Biosecurity Framework as Corporate Tool
The most provocative argument advanced by critics was that national security infrastructure, including DHS biosecurity authorities, could potentially be deployed to serve corporate agricultural interests rather than genuine public safety concerns. Under emergency pandemic declarations, authorities had broad powers to mandate medical interventions. Under agricultural biosecurity frameworks, similar powers could theoretically be applied to farming operations.
Whether these powers would ever be exercised in the manner critics feared remained speculative. But the legal architecture existed, and the pattern of regulatory alignment with industry interests lent the concerns a degree of plausibility that made them difficult to dismiss entirely.
Evaluating the Evidence
The debate over GMO safety, vaccine policy, and agricultural regulation involved a mixture of documented facts and speculative conclusions. The revolving door between Monsanto and regulatory agencies was a matter of public record. The reclassification of organic farming practices under biosecurity frameworks was documented in government directives. The CRIIGEN study, despite its methodological limitations, highlighted the absence of long-term feeding studies that should have been conducted before GMO crops entered the food supply.
At the same time, many of the more extreme claims in this debate lacked rigorous evidentiary support. The challenge for informed citizens was distinguishing between legitimate concerns about corporate influence and regulatory independence on one hand, and unfounded conspiratorial narratives on the other. The documented facts alone were sufficient to warrant serious scrutiny of the relationship between agricultural biotechnology companies and the government agencies tasked with protecting public health.